Quest for the right Drug
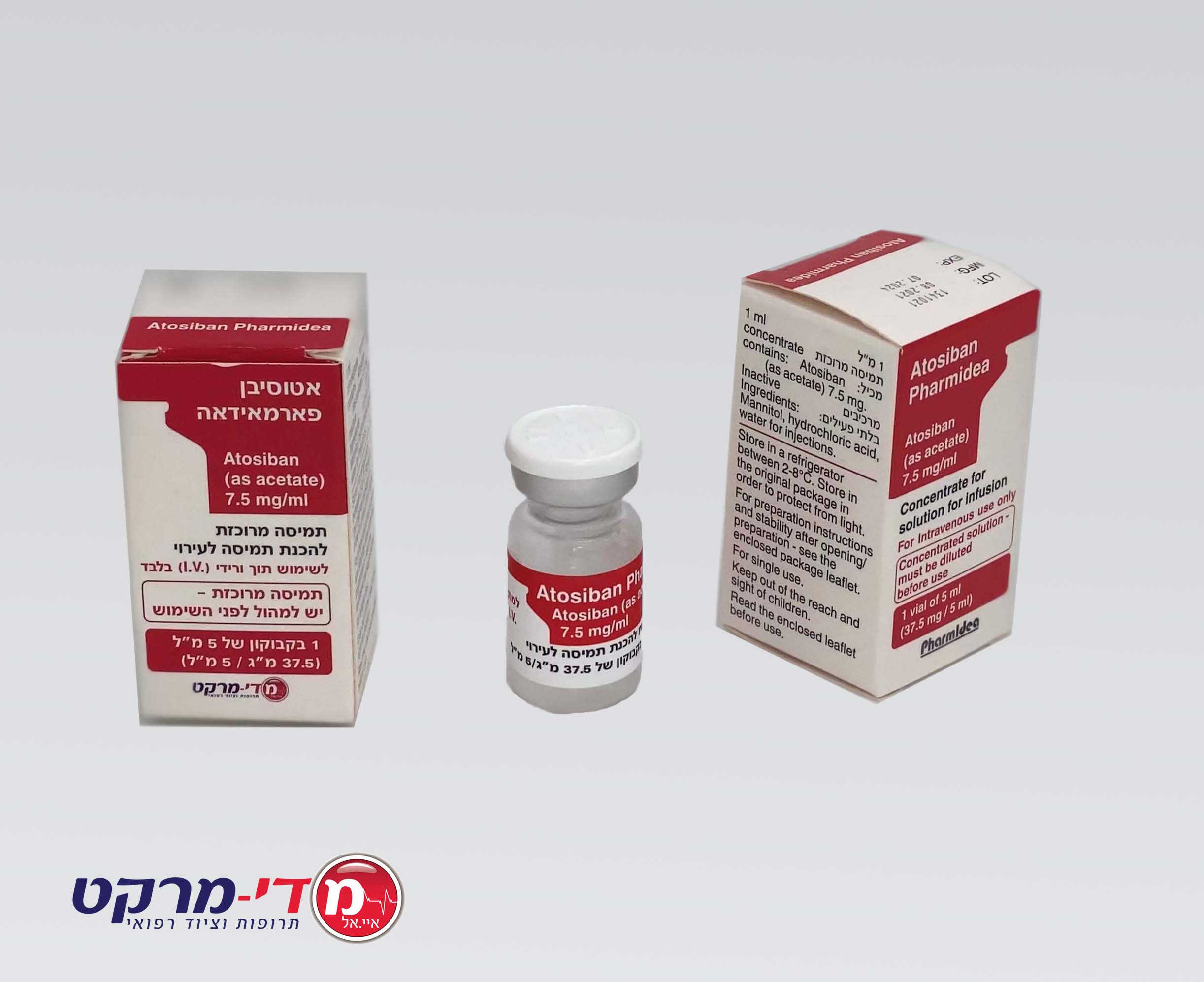
אטוסיבן פארמאידאה ATOSIBAN PHARMIDEA (ATOSIBAN AS ACETATE)
תרופה במרשם
תרופה בסל
נרקוטיקה
ציטוטוקסיקה
צורת מתן:
תוך-ורידי : I.V
צורת מינון:
אין פרטים : SOLUTION FOR INJECTION/ CONCENTRATE FOR SOLUTION FOR INFUSION
עלון לרופא
מינוניםPosology התוויות
Indications תופעות לוואי
Adverse reactions התוויות נגד
Contraindications אינטראקציות
Interactions מינון יתר
Overdose הריון/הנקה
Pregnancy & Lactation אוכלוסיות מיוחדות
Special populations תכונות פרמקולוגיות
Pharmacological properties מידע רוקחי
Pharmaceutical particulars אזהרת שימוש
Special Warning עלון לרופא
Physicians Leaflet
Adverse reactions : תופעות לוואי
4.8. Undesirable effects Possible adverse reactions of atosiban were described for the mother during the use of atosiban in clinical trials. In total 48% of the patients treated with atosiban experienced adverse reactions during the clinical trials. The observed adverse reactions were generally of a mild severity. The most commonly reported adverse reaction in the mother is nausea (14%). For the newborn, the clinical trials did not reveal any specific adverse reactions of atosiban. The infant adverse reactions were in the range of normal variation and were comparable with both placebo and beta-mimetic group incidences. The frequency of adverse reactions listed below is defined using the following convention: Very common (≥1/10); Common (≥1/100 to <1/10); Uncommon (≥1/1,000 to <1/100); Rare (≥1/10,000 to <1/1,000). Within each frequency grouping, adverse reactions are presented in order of decreasing seriousness. Very MedDRA System Organ Class (SOC) Common Uncommon Rare common Immune system disorders Allergic reaction Metabolism and nutrition disorders Hyperglycemia Psychiatric disorders Insomnia Headache Nervous system disorders Dizziness Cardiac disorders Tachycardia Vascular disorders Hypotention Hot flush Gastrointestinal disorders Nausea Vomiting Pruritis Skin and subcutaneous tissue disorders Rash Uterine haemorrage, Reproductive system and breast disorders Uterine atony General disorders and administration site conditions Injection site reaction Pyrexia Post-marketing experience Respiratory events like dyspnoea and pulmonary oedema, particularly in association with concomitant administration of other medicinal products with tocolytic activity, like calcium antagonists and beta-mimetics, and/or in women with multiple pregnancy, have been reported post-marketing. Reporting of suspected adverse reactions: adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Any suspected adverse events should be reported to the Ministry of Health according to the National Regulation by using an online form https://sideeffects.health.gov.il/

שימוש לפי פנקס קופ''ח כללית 1994
לא צוין
תאריך הכללה מקורי בסל
לא צוין
הגבלות
לא צוין
מידע נוסף
