Quest for the right Drug
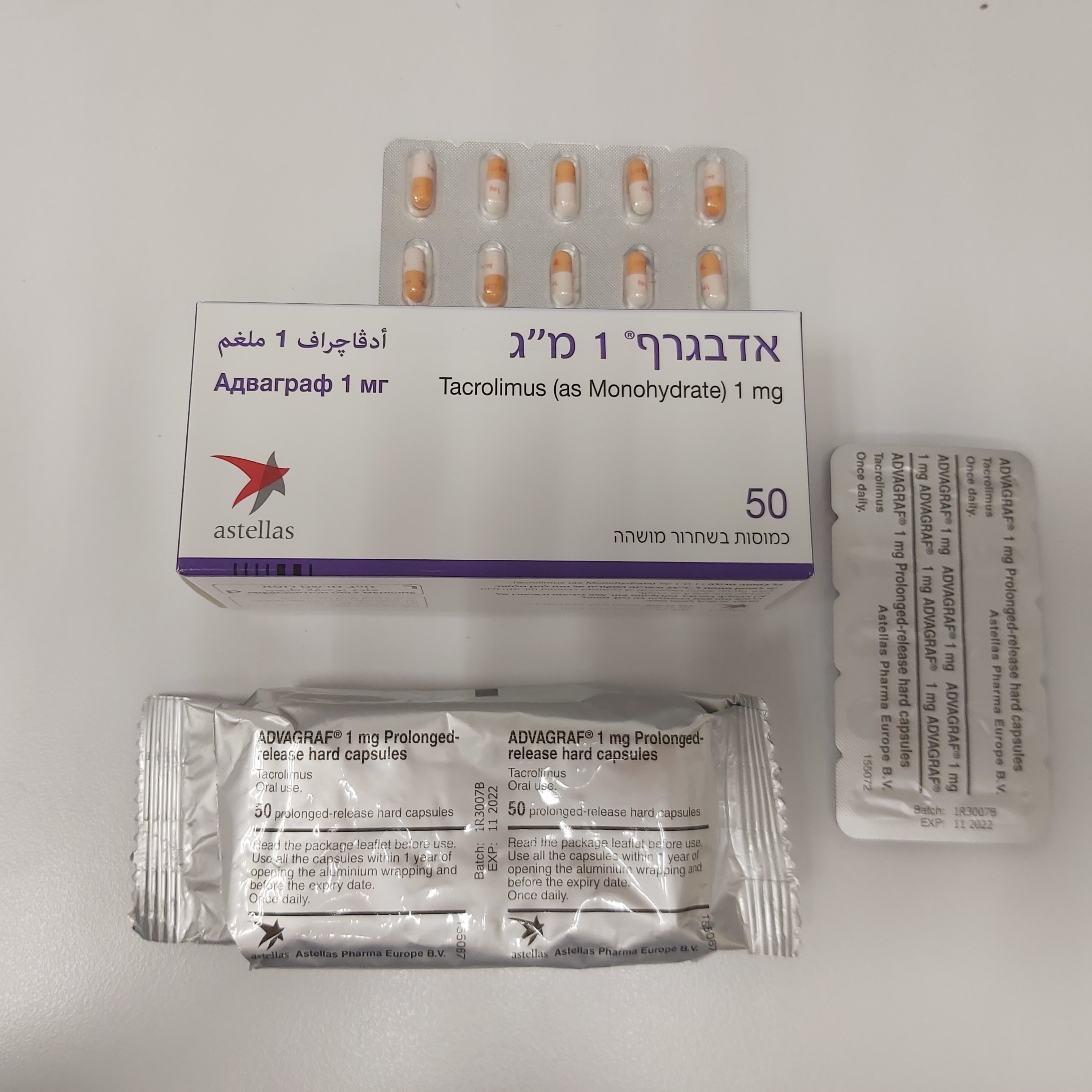
אדבגרף 1 מ"ג ADVAGRAF 1 MG (TACROLIMUS AS MONOHYDRATE)
תרופה במרשם
תרופה בסל
נרקוטיקה
ציטוטוקסיקה
צורת מתן:
פומי : PER OS
צורת מינון:
קפסולות בשחרור ממושך : CAPSULES PROLONGED RELEASE
עלון לרופא
מינוניםPosology התוויות
Indications תופעות לוואי
Adverse reactions התוויות נגד
Contraindications אינטראקציות
Interactions מינון יתר
Overdose הריון/הנקה
Pregnancy & Lactation אוכלוסיות מיוחדות
Special populations תכונות פרמקולוגיות
Pharmacological properties מידע רוקחי
Pharmaceutical particulars אזהרת שימוש
Special Warning עלון לרופא
Physicians Leaflet
Special Warning : אזהרת שימוש
4.4 Special warnings and precautions for use Medication errors, including inadvertent, unintentional or unsupervised substitution of immediate- or prolonged-release tacrolimus formulations, have been observed. This has led to serious adverse reactions, including graft rejection, or other adverse reactions which could be a consequence of either under- or over-exposure to tacrolimus. Patients should be maintained on a single formulation of tacrolimus with the corresponding daily dosing regimen; alterations in formulation or regimen should only take place under the close supervision of a transplant specialist (see sections 4.2 and 4.8). Advagraf is not recommended for use in children below 18 years due to limited data on safety and/or efficacy. For treatment of allograft rejection resistant to treatment with other immunosuppressive medicinal products in adult patients clinical data are not yet available for the prolonged-release formulation Advagraf. For prophylaxis of transplant rejection in adult heart allograft recipients clinical data are not yet available for Advagraf. During the initial post-transplant period, monitoring of the following parameters should be undertaken on a routine basis: blood pressure, ECG, neurological and visual status, fasting blood glucose levels, electrolytes (particularly potassium), liver and renal function tests, haematology parameters, coagulation values, and plasma protein determinations. If clinically relevant changes are seen, adjustments of the immunosuppressive regimen should be considered. Substances with potential for interaction Inhibitors or inducers of CYP3A4 should only be co-administered with tacrolimus after consulting a transplant specialist, due to the potential for drug interactions resulting in serious adverse reactions including rejection or toxicity (see section 4.5). CYP3A4 inhibitors Concomitant use with CYP3A4 inhibitors may increase tacrolimus blood levels, which could lead to serious adverse reactions, including nephrotoxicity, neurotoxicity and QT prolongation. It is recommended that concomitant use of strong CYP3A4 inhibitors (such as ritonavir, cobicistat, ketoconazole, itraconazole, posaconazole, voriconazole, telithromycin, clarithromycin or josamycin) with tacrolimus should be avoided. If unavoidable, tacrolimus blood levels should be monitored frequently, starting within the first few days of co-administration, under the supervision of a transplant specialist, to adjust the tacrolimus dose if appropriate in order to maintain similar tacrolimus exposure. Renal function, ECG including the QT interval, and the clinical condition of the patient should also be closely monitored. Dose adjustment needs to be based upon the individual situation of each patient. An immediate dose reduction at the time of treatment initiation may be required (see section 4.5). Similarly, discontinuation of CYP3A4 inhibitors may affect the rate of metabolism of tacrolimus, thereby leading to subtherapeutic blood levels of tacrolimus, and therefore requires close monitoring and supervision of a transplant specialist. CYP3A4 inducers Concomitant use with CYP3A4 inducers may decrease tacrolimus blood levels, potentially increasing the risk of transplant rejection. It is recommended that concomitant use of strong CYP3A4 inducers (such as rifampicin, phenytoin, carbamazepine) with tacrolimus should be avoided. If unavoidable, tacrolimus blood levels should be monitored frequently, starting within the first few days of co-administration, under the supervision of a transplant specialist, to adjust the tacrolimus dose if appropriate, in order to maintain similar tacrolimus exposure. Graft function should also be closely monitored (see section 4.5). Similarly, discontinuation of CYP3A4 inducers may affect the rate of metabolism of tacrolimus, thereby leading to supratherapeutic blood levels of tacrolimus, and therefore requires close monitoring and supervision of a transplant specialist. P-glycoprotein Caution should be observed when co-administering tacrolimus with drugs that inhibit P-glycoprotein, as an increase in tacrolimus levels may occur. Tacrolimus whole blood levels and the clinical condition of the patient should be monitored closely. An adjustment of the tacrolimus dose may be required (see section 4.5). Herbal preparations Herbal preparations containing St. John’s wort (Hypericum perforatum) or other herbal preparations should be avoided when taking Advagraf due to the risk of interactions that lead to either a decrease in blood concentrations of tacrolimus and reduced clinical effect of tacrolimus, or an increase in blood concentrations of tacrolimus and risk of tacrolimus toxicity (see section 4.5). Other interactions The combined administration of ciclosporin and tacrolimus should be avoided and care should be taken when administering tacrolimus to patients who have previously received ciclosporin (see sections 4.2 and 4.5). High potassium intake or potassium-sparing diuretics should be avoided (see section 4.5). Certain combinations of tacrolimus with drugs known to have neurotoxic effects may increase the risk of these effects (see section 4.5). Vaccination Immunosuppressants may affect the response to vaccination and vaccination during treatment with tacrolimus may be less effective. The use of live attenuated vaccines should be avoided. Nephrotoxicity Tacrolimus can result in renal function impairment in post-transplant patients. Acute renal impairment without active intervention may progress to chronic renal impairment. Patients with impaired renal function should be monitored closely as the dosage of tacrolimus may need to be reduced. The risk for nephrotoxicity may increase when tacrolimus is concomitantly administered with drugs associated with nephrotoxicity (see section 4.5). Concurrent use of tacrolimus with drugs known to have nephrotoxic effects should be avoided. When co-administration cannot be avoided, tacrolimus trough blood level and renal function should be monitored closely and dosage reduction should be considered if nephrotoxicity occurs. Gastrointestinal disorders Gastrointestinal perforation has been reported in patients treated with tacrolimus. As gastrointestinal perforation is a medically important event that may lead to a life-threatening or serious condition, adequate treatments should be considered immediately after suspected symptoms or signs occur. Since levels of tacrolimus in blood may significantly change during diarrhoea episodes, extra monitoring of tacrolimus concentrations is recommended during episodes of diarrhoea. Cardiac disorders Ventricular hypertrophy or hypertrophy of the septum, reported as cardiomyopathies, have been observed in Prograf treated patients on rare occasions and may also occur with Advagraf. Most cases have been reversible, occurring with tacrolimus blood trough concentrations much higher than the recommended maximum levels. Other factors observed to increase the risk of these clinical conditions included pre- existing heart disease, corticosteroid usage, hypertension, renal or hepatic dysfunction, infections, fluid overload, and oedema. Accordingly, high-risk patients receiving substantial immunosuppression should be monitored, using such procedures as echocardiography or ECG pre- and post-transplant (e.g., initially at 3 months and then at 9 -12 months). If abnormalities develop, dose reduction of Advagraf, or change of treatment to another immunosuppressive agent should be considered. Tacrolimus may prolong the QT interval and may cause Torsades de pointes. Caution should be exercised in patients with risk factors for QT prolongation, including patients with a personal or family history of QT prolongation, congestive heart failure, bradyarrhythmias and electrolyte abnormalities. Caution should also be exercised in patients diagnosed or suspected to have Congenital Long QT Syndrome or acquired QT prolongation or patients on concomitant medications known to prolong the QT interval, induce electrolyte abnormalities or known to increase tacrolimus exposure (see section 4.5). Lymphoproliferative disorders and malignancies Patients treated with tacrolimus have been reported to develop Epstein-Barr-Virus (EBV)-associated lymphoproliferative disorders (see section 4.8). A combination of immunosuppressives such as antilymphocytic antibodies (e.g., basiliximab, daclizumab) given concomitantly increases the risk of EBV-associated lymphoproliferative disorders. EBV-Viral Capsid Antigen (VCA)-negative patients have been reported to have an increased risk of developing lymphoproliferative disorders. Therefore, in this patient group, EBV-VCA serology should be ascertained before starting treatment with Advagraf. During treatment, careful monitoring with EBV-PCR is recommended. Positive EBV-PCR may persist for months and is per se not indicative of lymphoproliferative disease or lymphoma. As with other potent immunosuppressive compounds, the risk of secondary cancer is unknown (see section 4.8). As with other immunosuppressive agents, owing to the potential risk of malignant skin changes, exposure to sunlight and UV light should be limited by wearing protective clothing and using a sunscreen with a high protection factor. Infections including opportunistic infections Patients treated with immunosuppressants, including Advagraf are at increased risk for infections including opportunistic infections (bacterial, fungal, viral and protozoal) such as CMV infection, BK virus associated nephropathy and JC virus associated progressive multifocal leukoencephalopathy (PML). Patients are also at an increased risk of infections with viral hepatitis (for example, hepatitis B and C reactivation and de novo infection, as well as hepatitis E, which may become chronic). These infections are often related to a high total immunosuppressive burden and may lead to serious or fatal conditions including graft rejection that physicians should consider in the differential diagnosis in immunosuppressed patients with deteriorating hepatic or renal function or neurological symptoms. Prevention and management should be in accordance with appropriate clinical guidance. Posterior reversible encephalopathy syndrome (PRES) Patients treated with tacrolimus have been reported to develop posterior reversible encephalopathy syndrome (PRES). If patients taking tacrolimus present with symptoms indicating PRES such as headache, altered mental status, seizures, and visual disturbances, a radiological procedure (e.g., MRI) should be performed. If PRES is diagnosed, adequate blood pressure and seizure control and immediate discontinuation of systemic tacrolimus is advised. Most patients completely recover after appropriate measures are taken. Eye disorders Eye disorders, sometimes progressing to loss of vision, have been reported in patients treated with tacrolimus. Some cases have reported resolution on switching to alternative immunosuppression. Patients should be advised to report changes in visual acuity, changes in colour vision, blurred vision, or visual field defect, and in such cases, prompt evaluation is recommended with referral to an ophthalmologist as appropriate. Thrombotic microangiopathy (TMA) (including haemolytic uraemic syndrome (HUS) and thrombotic thrombocytopenic purpura (TTP)) The diagnosis of TMA, including thrombotic thrombocytopaenic purpura (TTP) and haemolytic uraemic syndrome (HUS), sometimes leading to renal failure or a fatal outcome, should be considered in patients presenting with haemolytic anaemia, thrombocytopenia, fatigue, fluctuating neurological manifestation, renal impairment, and fever. If TMA is diagnosed, prompt treatment is required, and discontinuation of tacrolimus should be considered at the discretion of the treating physician. The concomitant administration of tacrolimus with a mammalian target of rapamycin (mTOR) inhibitor (e.g., sirolimus, everolimus) may increase the risk of thrombotic microangiopathy (including haemolytic uraemic syndrome and thrombotic thrombocytopenic purpura). Pure Red Cell Aplasia Cases of pure red cell aplasia (PRCA) have been reported in patients treated with tacrolimus. All patients reported risk factors for PRCA such as parvovirus B19 infection, underlying disease or concomitant medications associated with PRCA. Special populations There is limited experience in non-Caucasian patients and patients at elevated immunological risk (e.g., retransplantation, evidence of panel reactive antibodies, PRA). Dose reduction may be necessary in patients with severe liver impairment (see section 4.2). Excipients As Advagraf capsules contain lactose, patients with rare hereditary problems of galactose intolerance, total lactase deficiency or glucose-galactose malabsorption should not take this medicine. The printing ink used to mark Advagraf capsules contains soya lecithin. In patients who are hypersensitive to peanut or soya, the risk and severity of hypersensitivity should be weighed against the benefit of using Advagraf. This medicine contains less than 1 mmol sodium (23 mg) per capsule, that is to say essentially ‘sodium-free’.
Effects on Driving
4.7 Effects on ability to drive and use machines Tacrolimus may cause visual and neurological disturbances. This effect may be enhanced if tacrolimus is administered in association with alcohol. No studies on the effects of tacrolimus (Advagraf) on the ability to drive and use machines have been performed.

פרטי מסגרת הכללה בסל
1. התרופה תינתן לטיפול במקרים האלה: א. מושתלי כליה ב. מושתלי כבד. ג. מושתלי לב. ד. מושתלי ריאה. 2. מתן התרופה ייעשה לפי מרשם של רופא מומחה באימונולוגיה קלינית או רופא מומחה העוסק בתחום ההשתלות
שימוש לפי פנקס קופ''ח כללית 1994
לא צוין
תאריך הכללה מקורי בסל
09/03/1999
הגבלות
תרופה מוגבלת לרישום ע'י רופא מומחה או הגבלה אחרת
רישום
153 08 34073 00
מחיר
0 ₪
מידע נוסף
עלון מידע לצרכן
26.04.22 - עלון לצרכן אנגלית 26.04.22 - עלון לצרכן עברית 24.05.20 - עלון לצרכן ערבית 05.12.22 - עלון לצרכן אנגלית 05.12.22 - עלון לצרכן עברית 05.12.22 - עלון לצרכן ערבית 25.08.15 - החמרה לעלון 05.08.18 - החמרה לעלון 06.09.20 - החמרה לעלון 26.04.22 - החמרה לעלון 05.12.22 - החמרה לעלוןלתרופה במאגר משרד הבריאות
אדבגרף 1 מ"ג