Quest for the right Drug
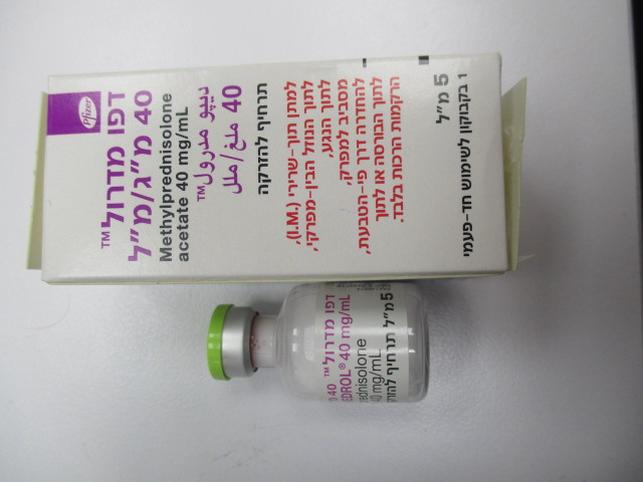
דפו מדרול 40 מ"ג/מ"ל DEPO MEDROL 40 MG/ML (METHYLPREDNISOLONE ACETATE)
תרופה במרשם
תרופה בסל
נרקוטיקה
ציטוטוקסיקה
צורת מתן:
זריקה ליד המפרק, תוך-שרירי, תוך מפרקי, לתוך פצע, לתוך נוזל העצם, להחדרה רקטלית, לרקמות רכות : PERIARTICULAR, I.M, INTRA-ARTICULAR, INTRA-LESIONAL, INTRABURSAL, INTRARECTAL INSTILLATION, SOFT TISSUES
צורת מינון:
תרחיף להזרקה : SUSPENSION FOR INJECTION
עלון לרופא
מינוניםPosology התוויות
Indications תופעות לוואי
Adverse reactions התוויות נגד
Contraindications אינטראקציות
Interactions מינון יתר
Overdose הריון/הנקה
Pregnancy & Lactation אוכלוסיות מיוחדות
Special populations תכונות פרמקולוגיות
Pharmacological properties מידע רוקחי
Pharmaceutical particulars אזהרת שימוש
Special Warning עלון לרופא
Physicians Leaflet
Interactions : אינטראקציות
4.5 Interaction with other medicinal products and other forms of interaction Methylprednisolone is a cytochrome P450 enzyme (CYP) substrate and is mainly metabolized by the CYP3A enzyme. CYP3A4 is the dominant enzyme of the most abundant CYP subfamily in the liver of adult humans. It catalyzes 6β-hydroxylation of steroids, the essential Phase I metabolic step for both endogenous and synthetic corticosteroids. Many other compounds are also substrates of CYP3A4, some of which (as well as other drugs) have been shown to alter glucocorticoid metabolism by induction (upregulation) or inhibition of the CYP3A4 enzyme. CYP3A4 INHIBITORS – Drugs that inhibit CYP3A4 activity generally decrease hepatic clearance and increase the plasma concentration of CYP3A4 substrate medications, such as methylprednisolone. In the presence of a CYP3A4 inhibitor, the dose of methylprednisolone may need to be titrated to avoid steroid toxicity. CYP3A4 INDUCERS – Drugs that induce CYP3A4 activity generally increase hepatic clearance, resulting in decreased plasma concentration of medications that are substrates for CYP3A4. Co-administration may require an increase in methylprednisolone dosage to achieve the desired result. CYP3A4 SUBSTRATES – In the presence of another CYP3A4 substrate, the hepatic clearance of methylprednisolone may be affected, with corresponding dosage adjustments required. It is possible that adverse events associated with the use of either drug alone may be more likely to occur with co-administration. 1. Convulsions have been reported with concurrent use of methylprednisolone and ciclosporin (CYP3A4 inhibitor and substrate). Since concurrent administration of these agents results in a mutual inhibition of metabolism (which may increase the plasma concentrations of either or both drugs), it is possible that convulsions and other adverse effects associated with the individual use of either drug may be more apt to occur. 2. Drugs that induce hepatic enzymes, such as rifampicin (antibiotic CYP3A4 inducer), rifabutin, carbamazepine (anticonvulsant CYP3A4 inducer and substrate), phenobarbitone and phenytoin (anticonvulsants CYP3A4 inducers), primidone, and aminoglutethimide (aromatase inhibitor) enhance the metabolism of corticosteroids and its therapeutic effects may be reduced. Aminoglutethimide- induced adrenal suppression may exacerbate endocrine changes caused by prolonged glucocorticoid treatment. The acetylation rate and clearance of isoniazid (CYP3A4 inhibitor), an antibacterial drug, can be increased by methylprednisolone. 3. Antibiotics/Antimycotics - Drugs such as erythromycin (macrolide antibacterial CYP3A4 inhibitor and substrate), itraconazole and ketoconazole (antifungal CYP3A4 2024-0093542 Page 13 of 21 inhibitors and substrates) may inhibit the metabolism of corticosteroids and thus decrease their clearance. Troleandomycin (CYP3A4 inhibitor), as well as clarithromycin, erythromycin, itraconazole and ketoconazole (CYP3A4 inhibitors and substrates) increase the effects and the side effects of methylprednisolone. 4. Steroids may reduce the effects of anticholinesterases in myasthenia gravis. The desired effects of hypoglycaemic agents (including insulin), anti-hypertensives and diuretics are antagonised by corticosteroids, and the hypokalaemic effects of acetazolamide, loop diuretics, thiazide diuretics and carbenoxolone are enhanced. An acute myopathy has been reported with the concomitant use of high doses of corticosteroids and anticholinergics, such as neuromuscular blocking drugs. (see section 4.4). Antagonism of the neuromuscular blocking effects of pancuronium and vecuronium has been reported in patients taking corticosteroids. This interaction may be expected with all competitive neuromuscular blockers. 5. The effect of methylprednisolone on oral anticoagulants is variable. The efficacy of coumarin anticoagulants may be enhanced by concurrent corticosteroid therapy and close monitoring of the INR or prothrombin time is required to avoid spontaneous bleeding and to maintain the desired anticoagulant effects. There are also reports of diminished effects of anticoagulants when given concurrently with corticosteroids. 6. There may be increased incidence of gastrointestinal bleeding and ulceration when corticosteroids are given with NSAIDs. Methylprednisolone may increase the clearance of high-dose aspirin, which can lead to decreased salicylate serum levels. Discontinuation of methylprednisolone treatment can lead to raised salicylate serum levels, which could lead to an increased risk of salicylate toxicity. Salicylates and non-steroidal anti-inflammatory agents should be used cautiously in conjunction with corticosteroids in hypothrombinaemia. 7. Antidiabetics- Because corticosteroids may increase blood glucose concentrations, dosage adjustments of antidiabetic agents may be required. 8. Antiemetics - Aprepitant and fosaprepitant (CYP3A4 inhibitors and substrates) 9. Antivirals - HIV protease inhibitors: 1) Indinavir ritonavir and pharmacokinetic enhancers (cobicistat) (CYP3A4 inhibitors and substrates) may increase plasma concentrations of corticosteroids. 2) Corticosteroids may induce the metabolism of HIV-protease inhibitors resulting in reduced plasma concentrations. 10. Calcium channel blocker - Diltiazem (CYP3A4 inhibitor and substrate). 11. Contraceptives (oral) - Ethinylestradiol/norethindrone (CYP3A4 inhibitors and substrate). 12. Other immunosuppressants like cyclophosphamide and tacrolimus are substrates of CYP3A4. 13. Potassium-depleting agents -When corticosteroids are administered concomitantly with potassium-depleting agents (e.g. diuretics), patients should be observed closely for development of hypokalaemia. There is also an increased risk of hypokalaemia 2024-0093542 Page 14 of 21 with concurrent use of corticosteroids with amphotericin B, xanthenes, or beta2 agonists. 14. Grapefruit juice – CYP3A4 inhibitor.

שימוש לפי פנקס קופ''ח כללית 1994
Rheumatoid arthritis, osteoarthritis, other arthritic conditions by intra-articular injection, inflammatory, allergic & rheumatic conditions requiring a glucocorticoid effect, in patients for whom treatment with oral corticosteroid is not feasible
תאריך הכללה מקורי בסל
01/01/1995
הגבלות
תרופה שאושרה לשימוש כללי בקופ'ח
מידע נוסף
עלון מידע לצרכן
24.10.12 - עלון לצרכן 02.05.16 - עלון לצרכן 16.08.18 - עלון לצרכן 18.12.18 - עלון לצרכן 25.07.22 - עלון לצרכן אנגלית 25.07.22 - עלון לצרכן עברית 25.07.22 - עלון לצרכן ערבית 23.06.23 - עלון לצרכן עברית 06.12.23 - עלון לצרכן אנגלית 05.12.23 - עלון לצרכן עברית 06.12.23 - עלון לצרכן ערבית 12.04.24 - עלון לצרכן עברית 05.09.24 - עלון לצרכן אנגלית 05.09.24 - עלון לצרכן ערבית 25.11.24 - עלון לצרכן עברית 10.09.20 - החמרה לעלון 19.01.21 - החמרה לעלון 25.10.21 - החמרה לעלון 21.09.23 - החמרה לעלון 12.04.24 - החמרה לעלון 08.07.24 - החמרה לעלון 25.11.24 - החמרה לעלוןלתרופה במאגר משרד הבריאות
דפו מדרול 40 מ"ג/מ"ל