Quest for the right Drug
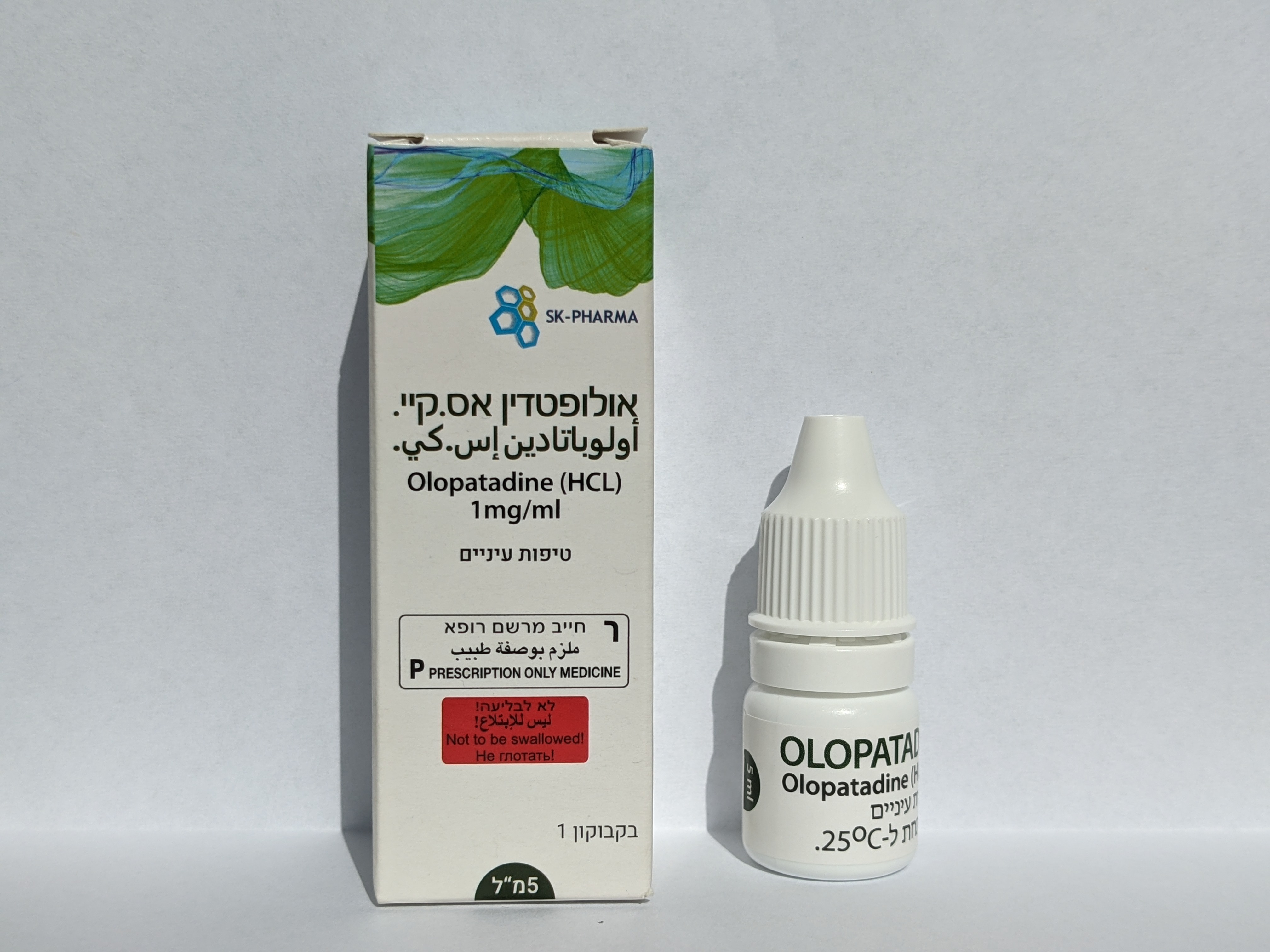
אולופטדין אס.קיי OLOPATADINE S.K. (OLOPATADINE AS HYDROCHLORIDE)
תרופה במרשם
תרופה בסל
נרקוטיקה
ציטוטוקסיקה
צורת מתן:
עיני : OCULAR
צורת מינון:
טיפות עיניים : EYE DROPS, SOLUTION
עלון לרופא
מינוניםPosology התוויות
Indications תופעות לוואי
Adverse reactions התוויות נגד
Contraindications אינטראקציות
Interactions מינון יתר
Overdose הריון/הנקה
Pregnancy & Lactation אוכלוסיות מיוחדות
Special populations תכונות פרמקולוגיות
Pharmacological properties מידע רוקחי
Pharmaceutical particulars אזהרת שימוש
Special Warning עלון לרופא
Physicians Leaflet
Pharmacological properties : תכונות פרמקולוגיות
Pharmacodynamic Properties
5.1 Pharmacodynamic properties Pharmacotherapeutic group: ophthalmologicals; decongestant and antiallergics; other antiallergics, ATC code: S01GX 09 Olopatadine is a potent selective antiallergic/antihistaminic agent that exerts its effects through multiple distinct mechanisms of action. It antagonises histamine (the primary mediator of allergic response in humans) and prevents histamine induced inflammatory cytokine production by human conjunctival epithelial cells. Data from in vitro studies suggest that it may act on human conjunctival mast cells to inhibit the release of pro-inflammatory mediators. In patients with patent nasolacrimal ducts, topical ocular administration of Olopatadine was suggested to reduce the nasal signs and symptoms that frequently accompany seasonal allergic conjunctivitis. It does not produce a clinically significant change in pupil diameter.
Pharmacokinetic Properties
5.2 Pharmacokinetic properties Absorption Olopatadine is absorbed systemically, as are other topically administered medicinal products. However, systemic absorption of topically applied olopatadine is minimal with plasma concentrations ranging from below the assay quantitation limit (<0.5 ng/ml) up to 1.3 ng/ml. These concentrations are 50-to 200-fold lower than those following well tolerated oral doses. Elimination From oral pharmacokinetic studies, the half-life of olopatadine in plasma was approximately eight to 12 hours, and elimination was predominantly through renal excretion. Approximately 60-70% of the dose was recovered in the urine as active substance. Two metabolites, the mono-desmethyl and the N-oxide, were detected at low concentrations in the urine. Since olopatadine is excreted in urine primarily as unchanged active substance, impairment of renal function alters the pharmacokinetics of olopatadine with peak plasma concentrations 2.3-fold greater in patients with severe renal impairment (mean creatinine clearance of 13.0 ml/min) compared to healthy adults. Following a 10 mg oral dose in patients undergoing haemodialysis (with no urinary output), plasma olopatadine concentrations were significantly lower on the haemodialysis day than on the non-haemodialysis day suggesting olopatadine can be removed by haemodialysis. Studies comparing the pharmacokinetics of 10 mg oral doses of olopatadine in young (mean age 21 years) and elderly (mean age 74 years) showed no significant differences in the plasma concentrations (AUC), protein binding or urinary excretion of unchanged parent drug and metabolites. A renal impairment study after oral dosing of olopatadine has been performed in patients with severe renal impairment. The results indicate that a somewhat higher plasma concentration can be expected with Olopatadine in this population. Since plasma concentrations following topical ocular dosing of olopatadine are 50-to 200-fold lower than after well- tolerated oral doses, dose adjustment is not expected to be necessary in the elderly or in the renally impaired population. Liver metabolism is a minor route of elimination. Dose adjustment is not expected to be necessary with hepatic impairment.

שימוש לפי פנקס קופ''ח כללית 1994
לא צוין
תאריך הכללה מקורי בסל
לא צוין
הגבלות
לא צוין
מידע נוסף
